Composite regenerative matrix for barrier-free bone regeneration: Full-mouth rehabilitation with long-term stability
Abstract
Keywords
Introduction
Materials and methods
Materials used
Freeze-dried bone allograft (FDBA)
Autologous fibrinogen glue (AFG)
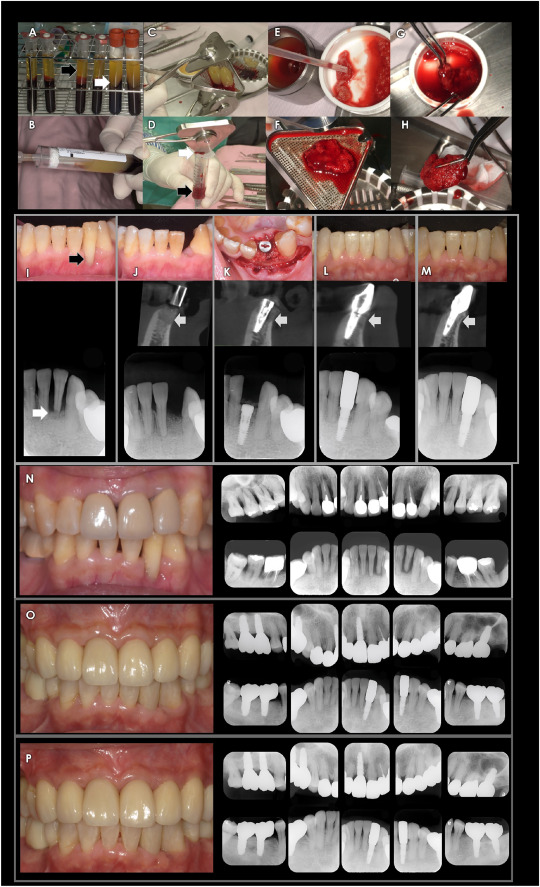
Figure 1. Preparation of the composite regenerative matrix (CRM) and treatment results. (A) Autologous blood was centrifuged using a concentrated growth factor (CGF) system with growth factor–rich autologous fibrinogen glue (AFG) in plastic tubes (white arrow) and coagulated CGF plugs in glass-coated tubes (black arrow). (B) Growth factor–rich AFG was aspirated from the plastic tube. (C) Coagulated CGF plugs obtained from glass-coated tubes were placed in a self-developed pressing device (D) upon closure of the self-developed device, and liquid autologous thrombin was expressed through the outlet (white arrow) and collected in a plastic syringe (black arrow). (E) Freeze-dried bone allograft (FDBA) particles were mixed with AFG in a sterile manipulation dish, and coagulation was initiated by adding autologous thrombin. (F) The composite matrix was subjected to controlled condensation using a self-developed condenser. (G) The condensed matrix was gently loosened in areas of weak integration supplemented with additional AFG and thrombin and allowed to recoagulate. (H) This compression and reapplication process was repeated several times to obtain a cohesive CRM mass suitable for defect filling. (I) Preoperative clinical photograph and radiograph of the mandibular left lateral incisor (tooth 32) region before extraction. (J) Four months after extraction and CRM-assisted socket regeneration, clinical findings and cone-beam computed tomography demonstrated adequate bone regeneration (white arrow). (K) Delayed implant placement with a gingival former was subsequently performed, and additional CRM (white arrow) was applied to support the peri-implant soft tissue contours. (L) The final prosthesis of tooth 32 was delivered in 2018. (M) More than six years of follow-up demonstrated stable peri-implant bone with well-modeled cortical bone located above the buccal implant platform (white arrow). (N) Full-mouth rehabilitation and long-term follow-up. Pretreatment clinical photographs and full-mouth periapical radiographs. (O) As part of a comprehensive treatment plan, similar regenerative procedures were performed at other sites within the same patient. After the completion of full-mouth rehabilitation, the definitive prostheses and radiographic outcomes were documented after three years in 2021. (P) At the four-year follow-up of full-mouth rehabilitation in 2025, clinical examination and full-mouth periapical radiographs confirmed stable hard and soft tissue conditions following reconstruction and regeneration.
Autologous thrombin
CRM preparation protocol
Initial mixing
Thrombin-induced coagulation
Primary compression
Loosening and reapplication
Secondary coagulation and compression
Ethical consideration
Results
Discussion
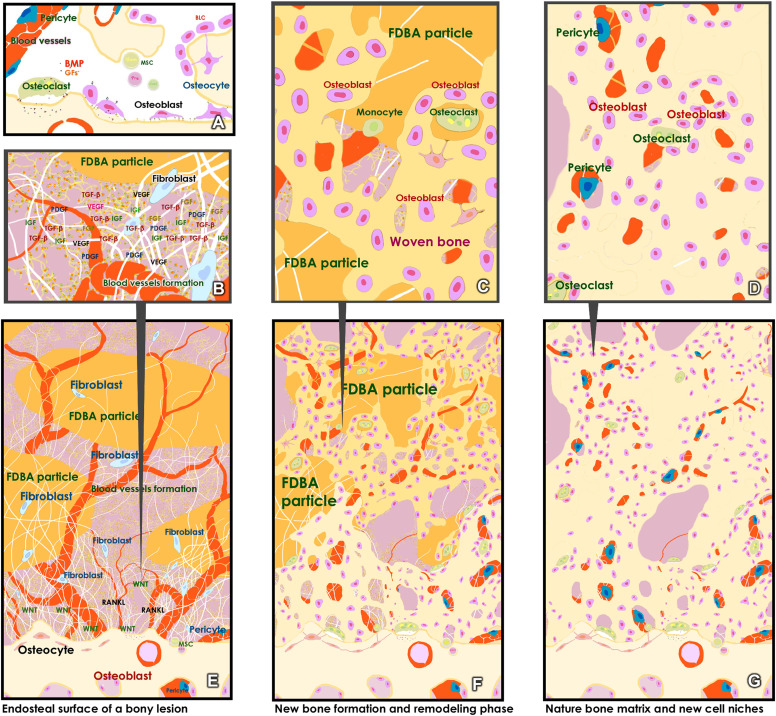
Figure 2. Conceptual schematic illustration of proposed biological events during the composite regenerative matrix (CRM)-assisted bone regeneration. (A) In a physiological bony environment, bone regeneration and remodeling are regulated by coordinated interactions among bone cells (osteoblasts, osteoclasts, osteocytes), their progenitors, the extracellular matrix (ECM), and local signaling cues. (B) Following the placement of a CRM, a fibrin-based scaffold incorporating freeze-dried bone allograft (FDBA) particles is proposed to provide a provisional matrix enriched with autologous blood-derived components. This matrix may support early vascular ingrowth and cellular migration within the defect space. (C) As regeneration progresses, osteoblast-mediated bone formation occurs in close association with the scaffold, resulting in the deposition of woven bone around residual graft particles. (D) Concurrently, osteoclasts derived from recruited mononuclear cells participate in bone remodeling by resorbing immature bone and graft material, contributing to matrix turnover and structural reorganization. (E and F) Through the coordinated bone formation and remodeling processes originating from both the endosteal and periosteal surfaces, the provisional matrix is gradually replaced by the newly formed bone with increasing mineral density and structural maturity. (G) Gradually, the continued remodeling results in a bone architecture resembling that of native alveolar bone with established cellular niches and vascular networks. This schematic represents a proposed biological sequence based on the current understanding of bone healing and is intended for illustrative purposes only.